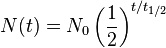Although I usually view the world on the macroscopic level, this physics chapter of nuclear radiation has got me thinking about the microscopic processes that are occurring in my daily lives. Of course I am not daily exposed to any dangerous radioactive emissions, but I do see the decay of carbon almost every day. However, I remember the specific time when I saw a decaying tree trunk on one of my family hikes at Manoa Falls.

Although they don’t know it, My sister and her husband stand before a partially decaying tree that is undergoing carbon decay
The tree trunk was cut down and laid on its side, left to rot for the next few years. By the looks of the soggy wood, we could tell that the trunk has already been decaying for a few years. At the time I was only interested in climbing the giant trunk that overlooked the rushing Manoa stream, but now after studying about the microscopic processes that occur during decay I can fully understand how the trunk was decaying at the atomic level.
Before the tree was cut down, it was alive and accepting the element nitrogen through the soil from a process called nitrification. The addition of the extra and free neutrons created carbon-14, one of the isotopes of carbon-12. In this process the a neutron is added to the nitrogen, already containing an atomic mass of 14. This addition causes the nitrogen to emit a proton, transmuting it to a carbon atom with the same atomic mass but a proton count of 6 instead of 7. The emitted proton and the added neutron create a hydrogen atom.
However, when the tree is dead it can no longer absorb nitrogen from the ground. Instead the once created carbon-14 begins to decay. The decay involves the carbon atom emitting a single electron, causing the atom to be slightly positive. Therefore, the atom will regain a proton returning to its nitrogen atom state. Over the years the amount of carbon will decrease until there is only a small amount remaining.
However, I could not see this process occurring during my time watching the massive decaying tree trunk, because this process, not only occurred at the atomic level, but it also occurred extremely slowly. The half-life for carbon decay is own to be 5,730 years. This means that when half of a sample’s carbon amount decays or when only half is left, 5,730 years would have elapsed. Then when half of the half of the sample would be left, the same amount of time would have elapsed. The process for all decaying matter is characterized by an exponential decay rate. The equation for finding the samples half-life also includes the initial and final amounts of the sample and the time that has already elapsed. Also another form of the equation gives the decay constant, that tells how much matter of the sample would be remaining after a given time. The equation is listed below:
Carbon-14 dating can be used to accurately date artifacts to millions of years old. However, since the tree trunk that I saw was only a few years into its decay process, it would be hard for the dating process to get a decent estimate on the trunk, because only a minute amount of carbon would have decayed. Still in a few thousand years carbon-14 dating could be effective in determining the age of the trunk.